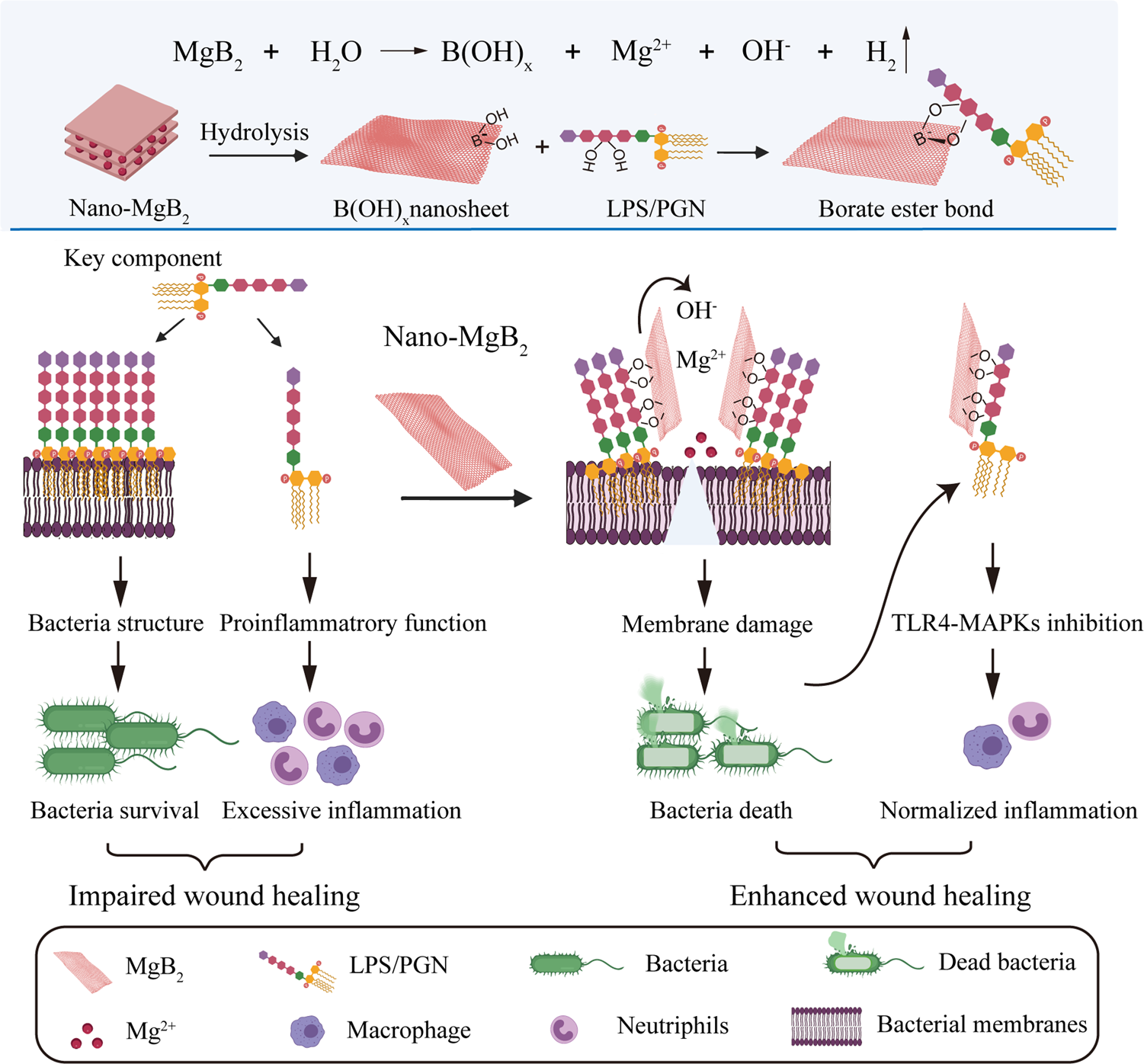
Structural basis to repurpose boron-based proteasome inhibitors Bortezomib and Ixazomib as β-lactamase inhibitors | Scientific Reports

Streamlined Process for the Chemical Synthesis of RNA Using 2′-O-Thionocarbamate-Protected Nucleoside Phosphoramidites in the Solid Phase | Journal of the American Chemical Society

Chemical structure of various blood antigens and antibody. a) O antigen... | Download Scientific Diagram
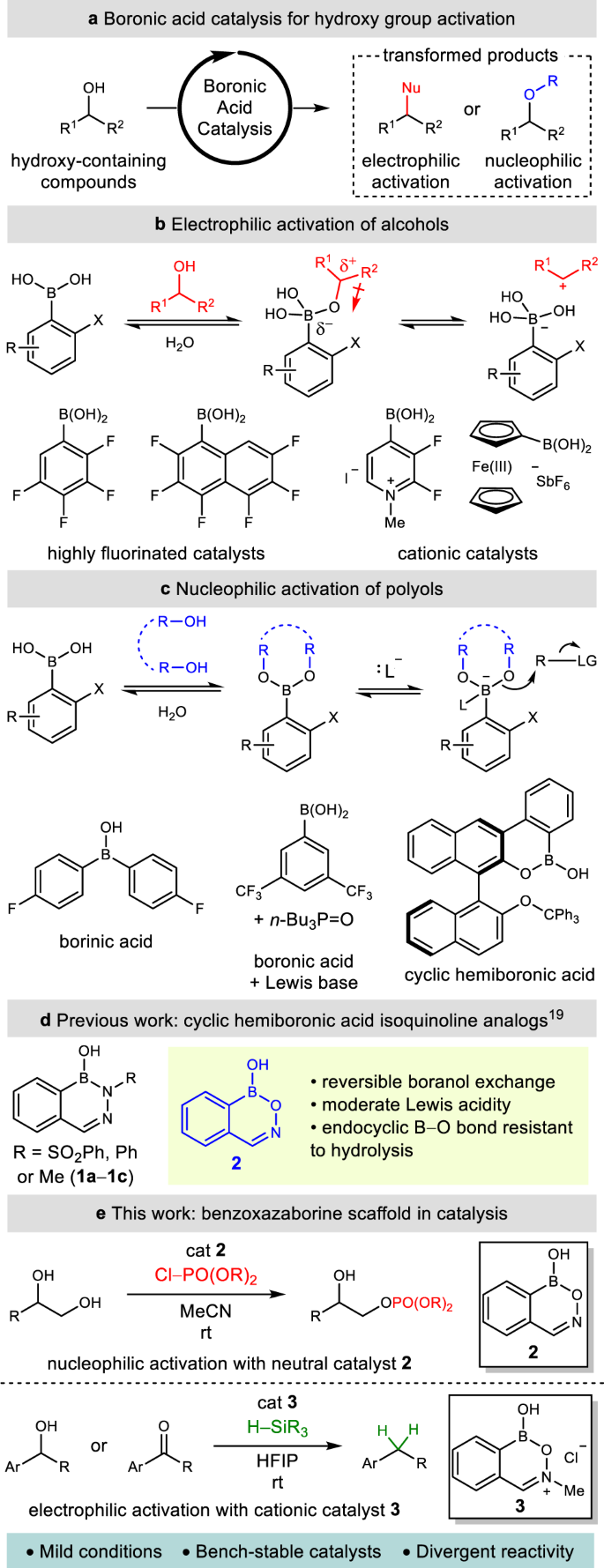
Direct nucleophilic and electrophilic activation of alcohols using a unified boron-based organocatalyst scaffold | Nature Communications
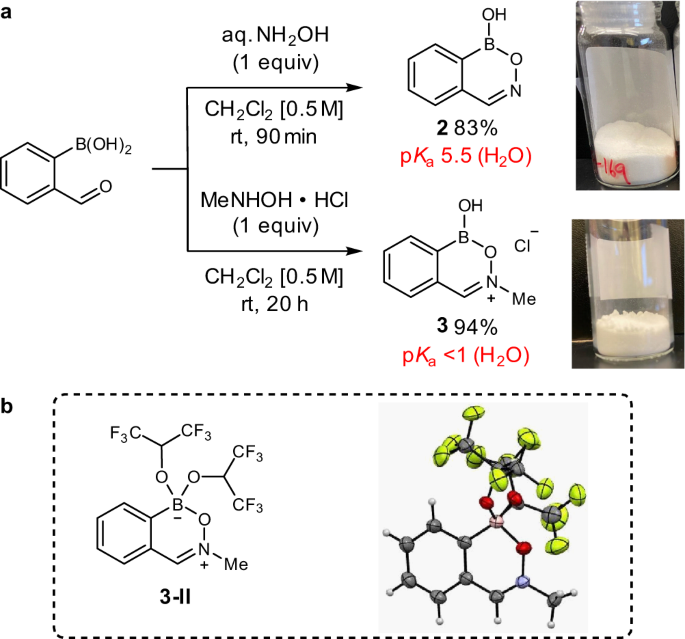
Direct nucleophilic and electrophilic activation of alcohols using a unified boron-based organocatalyst scaffold | Nature Communications

Tetrahydroxydiboron‐Mediated Palladium‐Catalyzed Deoxygenative Transfer Hydrogenation of Aryl Ketones - Spaller - 2022 - Advanced Synthesis & Catalysis - Wiley Online Library

B–N, B–O, and B–CN Bond Formation via Palladium-Catalyzed Cross-Coupling of B-Bromo-Carboranes | Journal of the American Chemical Society

Elucidating the Role of the Boronic Esters in the Suzuki–Miyaura Reaction: Structural, Kinetic, and Computational Investigations | Journal of the American Chemical Society

Tri(1-adamantyl)phosphine: Expanding the Boundary of Electron-Releasing Character Available to Organophosphorus Compounds | Journal of the American Chemical Society

Hydrogen peroxide-activatable antioxidant prodrug as a targeted therapeutic agent for ischemia-reperfusion injury | Scientific Reports
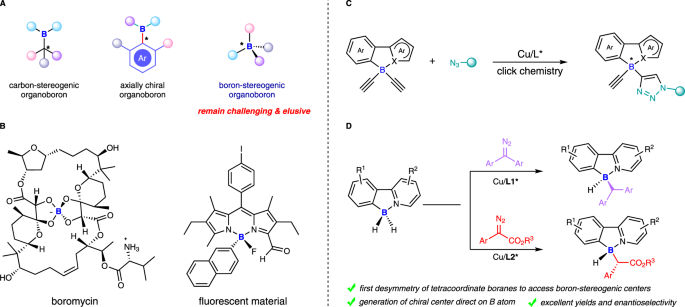
Construction of boron-stereogenic compounds via enantioselective Cu-catalyzed desymmetric B–H bond insertion reaction | Nature Communications

Effects of Fe Electrolyte Impurities on Ni(OH)2/NiOOH Structure and Oxygen Evolution Activity | The Journal of Physical Chemistry C